
The inner core electronic configuration of the initial state of the Pd is : This arises from spin-orbit coupling effects in the final state. The 3 d photoemission is in fact split between two peaks, one at 334.9 eV BE and the other at 340.2 eV BE, with an intensity ratio of 3:2. We can see this more clearly if, for example, we expand the spectrum in the region of the 3 d emission. the peak intensities are not simply related to the electron occupancy of the orbitalsĬloser inspection of the spectrum shows that emission from some levels (most obviously 3 p and 3 d ) does not give rise to a single photoemission peak, but a closely spaced doublet.there are significant differences in the natural widths of the various photoemission peaks.330 eV (in this case it is really meaningless to refer to an associated binding energy). the remaining peak is not an XPS peak at all ! - it is an Auger peak arising from x-ray induced Auger emission.335 eV is due to emission from the 3 d levels of the Pd atoms, whilst the 3 p and 3 s levels give rise to the peaks at ca.

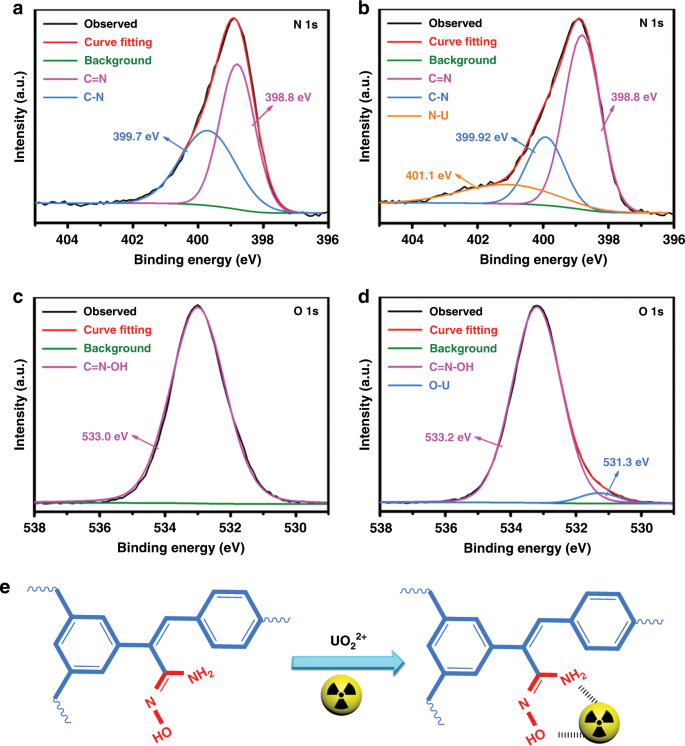
The 399-eV peak was attributed to the C−NH 2 or C−N bond, which was formed by electron transfer from the NO 2 molecule to the H-diamond surface and subsequent decomposition of NO 2 molecules at the H-diamond surface. Two nitrogen-related peaks were successfully detected by synchrotron radiation x-ray photoemission spectroscopy (XPS) at the interface of the Al 2O 3/NO 2/H-diamond structure, which is used for metal-oxide-semiconductor field-effect transistors.
